In both ESCs and differentiated cell lineages is critical to understanding the regulatory mechanisms used in maintaining and Quantitatively determining the complete transcriptional activity and the rate-limiting steps of transcription at all genes Moreover, expression of these core transcription factors is critical in programming the transition of differentiated mouseĮmbryonic fibroblasts (MEFs) to induced pluripotent stem cells, which have the properties and gene expression patterns ofĮSCs ( Mikkelsen et al. 2006 Rizzino 2008 Chambers and Tomlinson 2009). Perturbations to the transcriptional network established by these key transcription factors can result in a cascade ofĮvents that ultimately leads to the differentiation of stem cells ( Ivanova et al. These complexes localize to many promoters and regulate the transcription of target genes important for maintaining stemĬell identity ( Jaenisch and Young 2008 Kim et al. Network that is specific to pluripotent ESCs and early embryos ( Boyer et al. The core transcriptional factors OCT4, SOX2, and NANOG form a positive regulatory Pluripotency, and developmental progression. Mouse embryonic stem cells (ESCs) provide an excellent model for understanding the gene regulatory framework underlying self-renewal, Altogether, these findings identify rate-limiting targetsįor transcription regulation during cell differentiation. Pol II pausing, but it is confined to extremely 5′ proximal regions. In contrast, bivalent promoters bound by only PRC2 allow Levels of paused Pol II at promoters relative to an average gene. Histone modifications) bound by Polycomb group complexes PRC1 (Polycomb-repressive complex 1) and PRC2 show dramatically reduced Furthermore, “bivalent” ESC genes (exhibiting both active and repressive Importantly, quantitative comparisons between cell types reveal that transcription is controlledįrequently at paused Pol II's entry into elongation. Of a promoter-proximal, paused RNA polymerase II (Pol II) into productive elongation is a rate-limiting step in transcription RNA polymerases in mouse embryonic stem cells (ESCs) and mouse embryonic fibroblasts (MEFs). Profiles of RNA polymerases with high sensitivity and resolution, as well as the critical regulated steps upon which regulatoryįactors act, we used genome-wide nuclear run-on (GRO-seq) to map the density and orientation of transcriptionally engaged The genes and steps in the transcription cycle that are regulated during such transitions. Measurements of RNA polymerase distribution over the genome's primary transcription units in different cell states can identify Improving the sensitivity and throughput of these techniques.Transitions between pluripotent stem cells and differentiated cells are executed by key transcription regulators.Map transcription from all polymerases, including Pol I, Pol II, and Pol III.Determine transcription rates from time-course experiments.Determine immediate and directs transcriptional responses to various stimuli.Map TSSs of any transcript with high sensitivity.Map all transcripts regardless of the stability, including primary mRNAs, primary miRNAs, enhancer RNAs (eRNAs), RNAs that are upstream and antisense to promoters (uaRNAs), and transcription that continues beyond the 3’-ends of genes.GRO/PRO-cap: The methods above modified to map the sites of transcription initiation (Transcription Start Sites TSS). 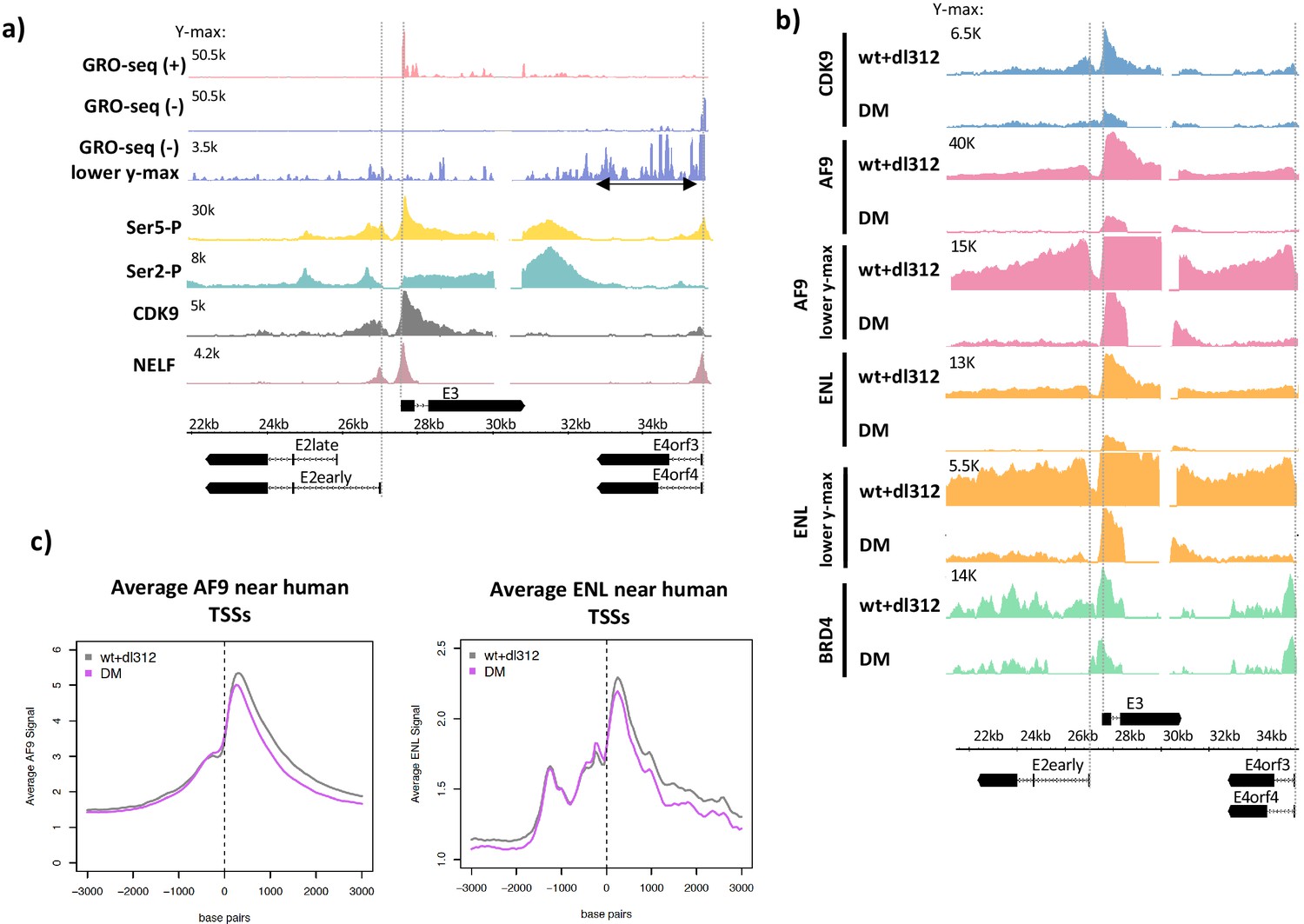
PRO-seq: Maps polymerase active site at near nucleotide resolution.GRO-seq: Maps polymerase location within 30-50bp.In previous work, we have developed nuclear run-on based assays that map where actively transcribing polymerase are in the genome by tracking the nascent RNA as it is being transcribed.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
